
The two pi bonds between carbon and nitrogen are formed due to the lateral overlap of two p orbitals (may be p x and p y or p x and p z or p y and p z). Thus, sigma bond is much stronger than pi bond. Sigma bond is the outcome of head on overlap of two atomic orbitals and pi bond is formed due to the lateral overlap of two atomic orbitals. Carbon is also bonded with one hydrogen atoms by one sigma bond. Among these three bonds, one is sigma bond and another two is pi bonds. Any covalent bonds is basically consisted of two electrons from each of the bond forming atoms.Ĭarbon is attached with nitrogen atom by three covalent bonds. Central atom is carbon and it is sp hybridized with a linear structure. To know more please follow: Is O2 a triple bond: Why, How, Characteristics and Detailed Facts HCN Sigma and Pi bondsįrom the point of hybridization the bond connectivity of central atom with each of the atom is clear. Hydrogen participates in the single bond formation with carbon by its one and only valance electrons. Similarly, nitrogen uses its three valance electrons among the five outer most shell electrons to form the triple bond with carbon and rest of the two valance electrons remain as nonbonded electron pair. Valance electron of hydrogen, carbon and nitrogen are 1, 4 and 5 respectively.Ĭentral atom, carbon uses its three valance electrons to form triple bond with nitrogen and last valance electron for the single bond formation with hydrogen atom. To determine the lewis structure of HCN, valance electrons of carbon, nitrogen and hydrogen should be counted from their electron configuration. Lewis structure or lewis dot structure helps to figure out the valance electrons or hybridization of any compound. To know more please check: 7 Tetrahedral Molecule Examples : Explanation And Detailed Facts HCN Lewis Structure C= Positive or negative charge of the molecule.GA= Groups of atoms attached to the central atom. 
Hybridization of HCN can be calculated using the following formula. One sp hybrid orbital from carbon atom combines with the 1s orbital of hydrogen atom and another sp hybrid orbital overlaps with one of the p orbitals among three p orbitals of nitrogen atom which are left as unhybridized or pure p-orbital. 2s and one 2p orbital of carbon atom participates in this hybridization. From this bond connectivity it is clear that carbon is sp hybridized and as a result hydrogen cyanide has a linear structure with a bond angle 180 0. In HCN, carbon is connected with nitrogen by triple bond and hydrogen with a single or sigma bond. The two bond forming atoms have one unpaired electron and after overlapping between the orbitals two unpaired electron from each of the atom get paired up. Two atoms share their half filled orbitals for overlapping to generate the hybrid orbitals. Like molecular orbital theory or MO theory, VBT also involved quantum mechanics.Īccording to this theory, bonding in any chemical species is the outcome of overlapping between the hybrid orbital of the consisting atoms. 
Chemical bonding between the atoms can be determined by VBT. 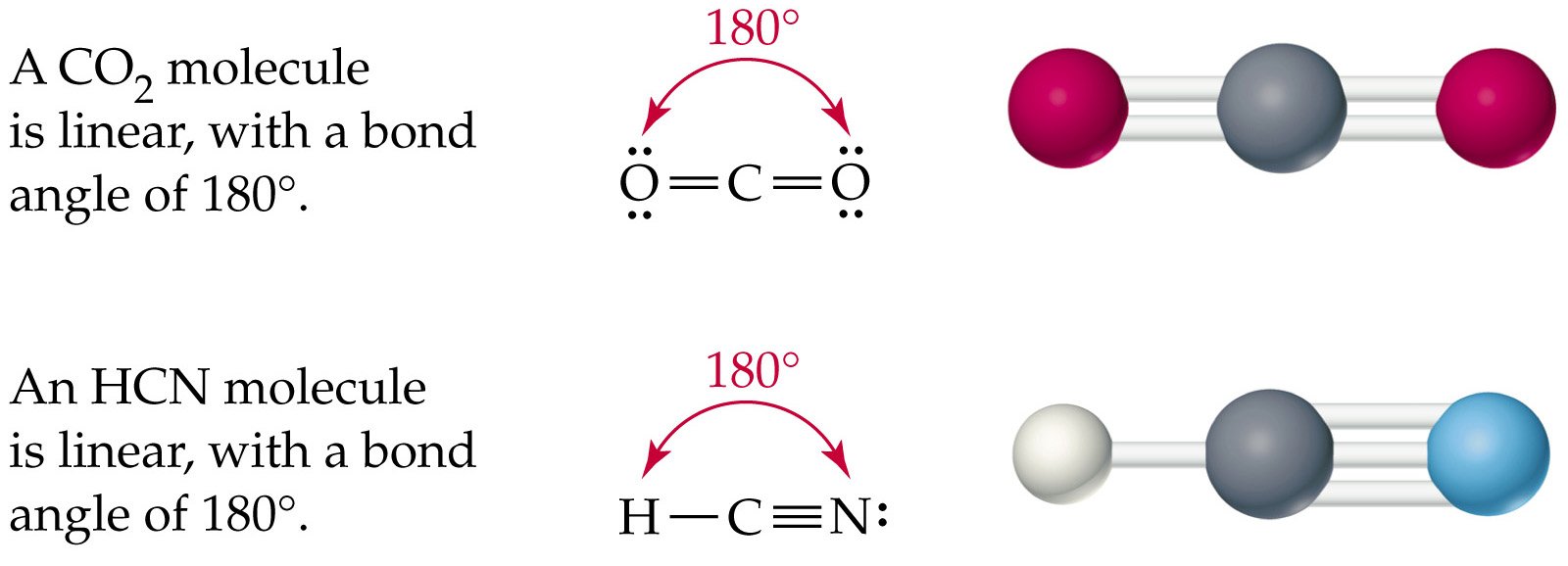
Hybridization of hydrogen cyanide can be easily explained by valance bond theory (VBT). In this chemical species, carbon is sp hybridized having bond connectivity with hydrogen and nitrogen by two sigma and two pi bonds.įew questions about the structure and hybridization of hydrogen cyanide are answered precisely below. Hydrogen cyanide known as prussic acid and it is a volatile, colorless and extremely toxic flammable liquid having a linear structure with a bond angle 180 0. In this article, “HCN hybridization”, hybridization, Lewis structure, bond connectivity of hydrogen cyanide with detailed explanation are discussed briefly.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
